International Journal of Scientific & Engineering Research, Volume 6, Issue 1, January-2015 1546
ISSN 2229-5518
Synthesis, characterization and biological evaluation of piperidine-4-carboxamide
derivatives in mice.
Saima Yaseen Baig, Shamim Akhter, Arfa Kamil, Lubna Bashir, Shazia Naz, Nausheen Alam, Ghazala Raza Naqvi, Tooba Siddiqi, Syeda Talat Naqvi, Mirza Tasawer Baig.
Abstract—Present work comprises of synthesis of sulfonamide and amide derivatives of piperidine-4-carboxamide via amino–de- chlorination and amino-de-alkoxylation reaction. Structure of the analogues was confirmed by different techniques i.e. IR and 1H NMR. Piperidine derivatives had found to be potent dopamine reuptake inhibitor. Synthesized derivatives were also shown to relieve pain and to achieve analgesia in mice. The antibacterial activity of the derivatives was also assessed with the parent against a series of Gram-positive and Gram-negative bacteria. The synthesized compounds showed diverse antimicrobial profile among which most compounds possessed a comparable or better activity in comparison to the parent.
Index Terms— Piperidine-4-carboxamide, dopamine reuptake inhibitor, analgesia, antibacterial activity.
1 INTRODUCTION
—————————— ——————————
IPERIDINE alkaloids constitute a large family of com- pounds many of which exhibit a wide range of physiolog- ical activities [1],[2]. These physiological activities of many
substituted piperidines prompted the scientists to design sim- ple methods for the synthesis of piperidine carboxamide de- rivatives.
Analgesic and anti-inflammatory drugs are the most common products used in many of disease for relief of pain and in- flammation. It has been found very difficult to treat chronic
pain using the treatments currently available. Therefore, the development of new analgesics has always been one of the main aim [3].Some 3-phenyl piperidine derivatives have shown significant analgesic activities. Some 1-amidino-3- amino-2-hydroxy piperidine derivatives were reported which possessed antiviral activities [4]. Advances in piperidine field are likely to provide better compounds capable of dealing with the resistant strains. These research efforts have been rewarded by very significant improvements in antibacterial potency as well as in vivo efficacy. In the light of above men- tioned briefly discussed research findings we prompted to prepare sulfonamide and amide derivative from piperidine molecule, by treating piperidine-4-carboxamide with 4- nitrobenzene sulfonyl chloride and methyl propionate respec- tively in order to assess the biological efficacy and behavioral activity. The present study enlightens the synthesis, spectro- scopic analysis (including IR and 1HNMR), and evaluation of behavioral and biological activities of piperidine-4- carboxamide derivatives in mice.
2 EXPERIMENTAL
2.1 Materials And Methods
Reactions were monitored by TLC using pre-coated silica gel, GF-254 and were visualized under ultraviolet light at 254nm and 360nm on HP UVIS Desaga (Heidelberg). Silica gel G 60 (0.040-0.063 mm) was also used for preparing analytical thin layer plates.
All melting points were recorded on Gallenkamp melting
point apparatus and were are uncorrected. Solid calcium sul-
phate (anhydrous) from E. Merck was used for drying metha- nol, ethanol and DMSO. U.V spectra were taken on spectro- photometer. Infra Red (IR) spectra were measured on a IR 460 spectrophotometer using KBr disc. Electron Impact Mass spec- tra (EIMS) were determined on Varain massen spectrometer. Proton Nuclear magnetic resonance (1HNMR) spectra were recorded in d6 -DMSO on Bruker AM-300 spectrometer operat- ing at 300 MHz. Chemical shifts (δ) were reported in parts per million (ppm) and coupling constant J in Hertz (Hz).
2.2 General Procedure For Preparation Of Derivatives Piperidine-4-carboxamide, 4-nitrobenzene sulfonyl chloride and methyl propionate were dissolved in acetone in equimolar
quantities separately in conical flasks and then mixed together
in a round bottom flask. The reaction mixtures were stirred by
magnetic stirrer and refluxed for about 4 to 5 hours. Comple- tion of the reactions were monitored by TLC with CHCl3 - MeOH (in varying proportions) system. The resulting precipi- tates of products were filtered and washed with warm acetone to remove the unreacted starting materials. The products thus obtained were recrystallized with pure ethanol. The pure
IJSER © 2015 http://www.ijser.org
International Journal of Scientific & Engineering Research, Volume 6, Issue 1, January-2015 1547
ISSN 2229-5518
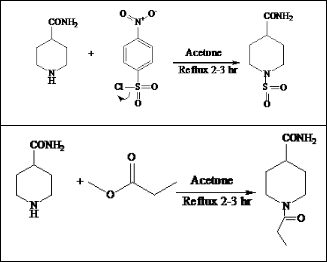
compounds were dried in vaccum desiccator over anhydrous calcium sulphate.
єmax : 539, IRν max(KBr)cm-1 3384, 2947, 2792, 2914, 2617, 2520,
1651, 1450, 1365, 1299, 1342, EIMS:m/z: 216 (C10 H20 N2 O3 ), 141,
100, 94, 73, 72, 70, 71, 57, 56, 551H NMR (d6- DMSO, 300 MHz)δ:

2.03(2H, m, H-5), 2.33(2H, m, H-3), 2.9(1H, m, H-4) 3.03 (2H, m, H-6), 3.33 (2H, m, H-2), 1.86( 1H, t, H-7, J=12.18 Hz, J=23.26 Hz), 2.1(2H, m, H-8), 2.7 (3H, s, H-9), 3.43(3H, s, OCH3 , H-10)CHN Anal (C10 H20 N2 O3 ): Calculated (%):

216.28, C-55.418, H-9.221, N-12.95 Found:C-55.6, H-
9.26, N-12.165
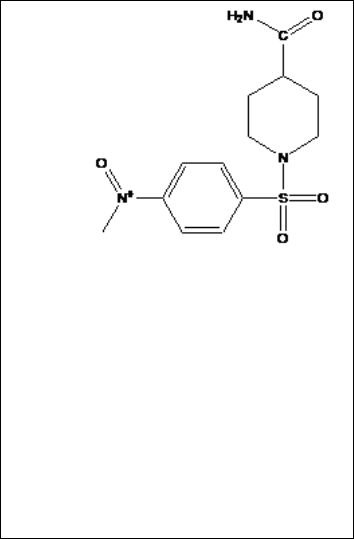
1-(4-Nitro-benzene sulfonyl piperidine-4-Carboxylic acid
amide
S(S4)
2.3 Characterization of synthesized compounds and
Physical Data :
The structures of synthesized compounds were elucidated by I
UV, IR, EIMS and Proton NMR spectral techniques. The spec-
tral and physical data are given as under. ,
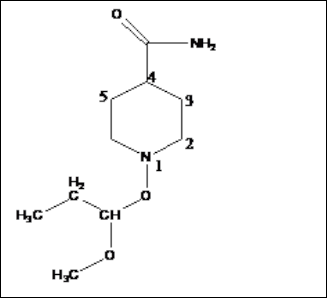
1-(1-Methoxy-propoxy)-piperidine-4-Carboxylic acid amide
(SS2)
Colour and physical state: White powder Molecular
Formula: C12H15N3O5S, Molecular Weight: 349.8
Melting Point: 237°C, Yield:52% , UV λmax: 270nm єmax:
8380
IR ν max(KBr)cm-1:3404, 3199.7, 2939, 2887, 1637.5, 1541,
1350, 1057, EIMS m/z: 349.80(C12H15N3O5S), 127.1, 82.0,
122.0, 83, 75, 72, 56, 55, 128.1.1H-NMR (d6-DMSO, 300
MHz):δ 2.03(2H, m, H-5), 2.23(2H,m,H-3), 2.9 (1-H, m, H-4),
3.03(2H, m, H-6), 3.33 (2H, m, H-2), 1.86(1H, t, H-7, J=12.18
Hz, J=23.226 Hz), 2.1(2H,
m, H-8), 2.7(3H, s, H-9), 3.43(3H, s, OCH3, H-10), 8.04-
8.07(dd, J=1.839 Hz, J=8.805 Hz, 2H, H-7, H-10), 8.4-8.48(dd, J=2.169 Hz, J =8.884 Hz, 2H, H-8,H-9)CHN Analysis (C12H15N3O5S): Calculated (%): C-41.17, H-4.2, N-12.0
Found:C-42.1, H-4.6, N-12.1
Color and physical state: Off white powder Molecular
Formula: C10 H22 N2 O3, Molecular Weight: 216
————————————————
• Corresponding author: Dr.Nausheen Alam
• E-mail:nausheenasaosh@hotmail.com
Melting Point: 170°C , Yield: 49%, UVλmax: 220nm
2.4 Methodology of Pharmacological Evaluations
Analgesic activity
This activity was carried out on white Albino mice of either
sex (locally bred) weighing between 20-30 gm. The
compounds were tested for their analgesic activity as
antinociceptive effect against thermal stimuli (tail flick
method)[5]. Groups of five animals were maintained under
standard 12 hours light/12 hours dark at temperature 25±1°C
IJSER © 2015 http://www.ijser.org
International Journal of Scientific & Engineering Research, Volume 6, Issue 1, January-2015 1548
ISSN 2229-5518
(room temperature), fed with frequent diet and water. Derivatives were dissolved in water and injected to the test animals intraperitoneally at the doses of 50mg/kg body
maximum in 120 minutes and effect lasted upto 180 minutes showing long duration of action. It is also interesting to note that it seems to be more potent as compared to pethidine
administration of test compounds and standard drugs (0 minutes) and then 30, 60, 90, 120, 150 and 180 minutes after the administration of compounds. The criteria of analgesia was the difference in post drug and pre drug latency which was greater than two times the pre drug average latency[6]. Mean increase in latency after drug administration or Analgesia TFLD was calculated as follows:
Analgesia TFLD = Post drug tail flick latency - pre drug tail flick latency)
Analgesic activity was expressed as TFLD ± SEM in terms of seconds. Statistical analysis was performed using student t- test and values were considered significant or highly signifi- cant, P<0.05 or P<0.01 respectively. All statistical procedures
Fig. 1.
-4-
Behavioral Studies
Compounds were dissolved in water for injection and injected
to the test animals intraperitoneally (i.p.) at the dose of
50mg/Kg body weight. Water for injection was injected by the
same route to the control animals. The open field apparatus
consisted of a square area 24 x 24 cm with 14 cm height. Floor
of the apparatus was divided by lines into 25 equal squares.
The mice were exposed to the open field after 30 minutes after
receiving injection. The activity was scored as number of
squares crossings with all four paws for 5 minutes.
Antimicrobial Screening
The test compounds were evaluated for the antimicrobial ac-
tivity by disc diffusion method[8]. Different Gram negative
and Gram positive organisms were seeded over previously
sterilized nutrient agar. The zones of inhibition were measured
around the dried discs of Whattman containing l0µl of the test
sample. Blank discs containing DMSO were used as control.
The plates were incubated at 37°C for 24 hours.
3. Result and Discussion
The antinociceptive test used in this work is chosen to test nociceptive stimuli namely cutaneous thermic (thermal) which is the characteristic of central narcotic analgesics. An increase in the central inhibitory neurotransmitter, γ-aminobutyric acid (GABA) explains the antinociceptive effects [9]

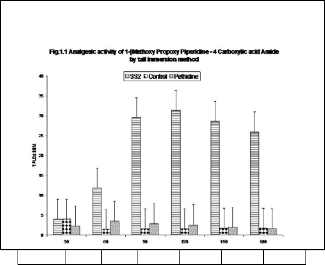
Tail-flick response is essentially a spinal reflex[10]. It has al- ready been shown that tail-flick method is specifically used for screening the strong narcotic effects [11]. Involvement of µ and κ-opioid receptor can also be suggested[12]. The effect on tail- flick response provides a confirmation of this central effect since, the assay is specific for opioid induced antinociceptive effect (9).Results of antinociceptive effects of substituted de- rivatives of piperidine-4-carboxamide were shown in the ta- bles 1and 2 showing the analgesia of varying degrees and its duration by tail immersion method [13]. Result shows the ef- fects produced by the compound SS2 demonstrated highly significant effects. The onset of action was early reaches to
HCl 0.63 0.46 0.13 0.23 0.07 0.20
Significant difference by student’t test when *p<0.05 and highly significant when **p<0.01 as compared to control n /group = 5
Table – 2:Analgesic Effect of 1-(4-Nitrobenzene-sulfonyl)
piperidine-4-carboxylic acid amide (SS4)
Treat- ment (IP) | Mean Increase in Latency after drug administration ± SEM |
Dose (50mg/ kg) | 30 min | 60 min | 90 min | 120 min | 150 min | 180 min |
Con- trol | 1.52 ± 0.6 | 1.56 ± 1.62 | 1.63 ± 1.65 | 1.65 ± 1.66 | 1.68 ± 1.69 | 1.7 ± 1.69 |
SS4 | 4.82 ** ± 1.04 7 | 14.6 4** ± 2.30 | 31.53 ** ± 7.04 | 28.91 ** ± 5.29 | 28.62 ** ± 6.40 | 25.18 ** ± 5.39 |
Pethi- dine HCl | 2.26 * ± 0.63 | 3.52 ** ± 0.46 | 2.82* * ± 0.13 | 2.57* * ± 0.23 | 1.92* * ± 0.07 | 1.57* * ± 0.20 |
Significant difference by student’t test when *p<0.05 and highly sig- nificant when **p<0.01 as compared to control n / group = 5
Table: 3: Effect of Piperidine-4-Carboxamide Derivatives
(SS2&SS4) on behavior in Open Field Test in mice
Treatment (IP)Dose (50 mg/Kg) | Number of squares cross | t-test | Latency to move (sec) | t-test |
Control | 38 ± 1.91 | | 1 ± 0 | |
SS2 | 75.42** ± 0.54 | 3.909 P<0.01 | 21.8** ± 21.04 | 5.509 P<o.o1 |
SS4 | 2.54** ± 1.307 | 1.428 P<0.01 | 0.31 ± 1.29 | 0.781 P<0.01 |
Values are mean ± SD. (n=5), 30 minutes after injection. Significant differences by student t test when *p<0.05, and highly significant when **p>0.01 as compared to control.
IJSER © 2015 http://www.ijser.org
International Journal of Scientific & Engineering Research, Volume 6, Issue 1, January-2015 1549
ISSN 2229-5518
Results of compound SS4 also demonstrated highly signifi-
Table – 4: Antibacterial Activity of synthetic compounds against gram-positive organisms and their zones of inhibition in mm.
Synthet- ic com- pound | Name of bacterial species (gram-positive) |
| Bac- sub- tilis | Bac- cere- us | Staph - aure- us | Staph- epider- midis | Corynebacte- rium diiphtherae |
SS2 | 12 | 12 | 10 | - | - |
SS42 | 1412 | 12 | 10 | 10 | 10 |
Activity key=No of zone of inhibition=14=Better activi-
ty=12=good activity=10=moderate activity
Fig. 2

Table – 5 Antibacterial Activity of synthetic compounds against gram-negative organisms and their zones of in- hibition in mm.
Fig. 1.2. Analgesia Produced by-1-(Nitro-Benzene Sulfonyl Piperidine -4 Carboxylic acid amide by Tail immersion method
40 SS4 Control Pethidine
35
30
25
20
15
10
5
0
Fig. 3
30 60 90 120 150 180
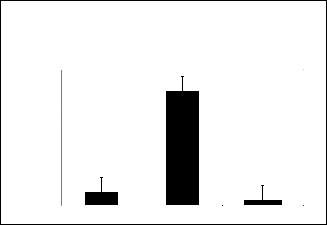
Fig.1.3. Effect of Piperidine -4 Carboxamide Derivatives (SS2 and SS4) on behavior in open field Test in mice
cant analgesic effects. Again the onset of action is fast and the duration is longer lasting. It is shown that analgesia started within 15 minutes, reaches to maximum in 90 minutes and decreasing slowly but still highly significant effects can be seen upto 180 minutes.
It is well established that locomotor activity can show positive effects in the antidepressant tests [14]. Therefore, a precise method was established to determine the effect on the mobili- ty and the open field activity produced by the synthesized compounds. Open field activity of the newly synthesized de- rivatives SS2 and SS4 are presented in table 3.
It has already been studied in our synthetic lab that the parent
90
compound Piperidine-4-carboxamide was devoid of hyper or
80
hypomotility [15] while the synthesized compounds possessed
60 significant activity. The compound SS2, produced hypermotil-
50 ity in animals and showed highly significant activity (P<0.01)
40 at the dose of 50mg/kg body weight depicted by the number
30 of squares crossed. Whereas, the compound SS4 produced
20
10
0
Control SS2 SS4
Time
hypomotility in the animals.
The primary mechanism underlying behavioral effects includ-
ing locomotion stimulation was thought to be due to its ability to bind to dopamine receptors [16]. In agreement with the do- pamine hypothesis, piperidine derivatives had been found to inhibit dopamine reuptake and motor effects. It can also be suggested that besides the inhibition of dopamine reuptake,
IJSER © 2015 http://www.ijser.org
International Journal of Scientific & Engineering Research, Volume 6, Issue 1, January-2015 1550
ISSN 2229-5518
other mechanisms might also play a modulatory role in in- creasing the locomotion in animals.
Antibacterial activity of compound against Gram positive or- ganisms SS4 appeared more effective as compared to SS2, as observed against Bacillus sp. with 14 mm zone of inhibition and other bacteria including Bacillus cereus, S. aureus, S. epi- dermidis & C. diphtherae were also inhibited by SS4, It has shown 12 mm &10 mm zones of inhibition respectively. Over all activity of SS4 against gram positives is good as compared to SS2. SS2 has shown 12 mm to 10 mm zone of inhibition against Bacillus subtillus, B. cereus and S. aureus respectively. Antibacterial activities of test samples against Gram negatives indicate that S. typhi is the only susceptible organism among all tested strains. Both synthetic compounds SS2 and SS4 have indicated same antibacterial potential against gram negatives
4 CONCLUSION
It can be concluded that if the study proceeded further, the duration of action could be determined. This long duration shows that drug was not metabolized upto 180 minutes or it may be possible that the metabolized components might also responsible to produce analgesia. Hypothesis, piperidine derivatives had been found to inhibit dopamine reuptake and motor effects. It can also be suggested that besides the inhibi- tion of dopamine reuptake, other mechanisms might also play a modulatory role in increasing the locomotion in animals. A complete overview of the results of Pharmacological and Bio- logical activities of both of these novel derivatives clearly demonstrates that both of them could be proved as most ac- tive molecules, although further exploration of these com- pounds is required to make them safe and potent therapeutic agents.
REFERENCES
[1] J,W Daly, Spande T.F., H M Garrafo J. Nat. Product 68, 1556 (2005).
[2] W.-K. Chen, Linear Networks and Systems. Belmont, Calif.: Wadsworth, pp. 123-135, 1993. (Book style)
[3] H. Poor, “A Hypertext History of Multiuser Dimensions,” MUD His- tory, http://www.ccs.neu.edu/home/pb/mud-history.html. 1986. (URL link *include year)
[4] K. Elissa, “An Overview of Decision Theory," unpublished. (Unplu- blished manuscript)
[5] R. Nicole, "The Last Word on Decision Theory," J. Computer Vision,
submitted for publication. (Pending publication)
[6] C. J. Kaufman, Rocky Mountain Research Laboratories, Boulder, Colo., personal communication, 1992. (Personal communication)
[7] D.S. Coming and O.G. Staadt, "Velocity-Aligned Discrete Oriented Polytopes for Dynamic Collision Detection," IEEE Trans. Visualization and Computer Graphics, vol. 14, no. 1, pp. 1-12, Jan/Feb 2008, doi:10.1109/TVCG.2007.70405. (IEEE Transactions )
[8] S.P. Bingulac, “On the Compatibility of Adaptive Controllers,” Proc.
Fourth Ann. Allerton Conf. Circuits and Systems Theory, pp. 8-16, 1994. (Conference proceedings)
[9] H. Goto, Y. Hasegawa, and M. Tanaka, “Efficient Scheduling Focus- ing on the Duality of MPL Representation,” Proc. IEEE Symp. Compu- tational Intelligence in Scheduling (SCIS ’07), pp. 57-64, Apr. 2007, doi:10.1109/SCIS.2007.367670. (Conference proceedings)
[10] J. Williams, “Narrow-Band Analyzer,” PhD dissertation, Dept. of Elec- trical Eng., Harvard Univ., Cambridge, Mass., 1993. (Thesis or disserta- tion)
[11] E.E. Reber, R.L. Michell, and C.J. Carter, “Oxygen Absorption in the Earth’s Atmosphere,” Technical Report TR-0200 (420-46)-3, Aerospace Corp., Los Angeles, Calif., Nov. 1988. (Technical report with report number)
[12] L. Hubert and P. Arabie, “Comparing Partitions,” J. Classification, vol.
2, no. 4, pp. 193-218, Apr. 1985. (Journal or magazine citation)
[13] R.J. Vidmar, “On the Use of Atmospheric Plasmas as Electromagnetic Re- flectors,” IEEE Trans. Plasma Science, vol. 21, no. 3, pp. 876-880, available at http://www.halcyon.com/pub/journals/21ps03-vidmar, Aug. 1992. (URL for Transaction, journal, or magzine)
[14] J.M.P. Martinez, R.B. Llavori, M.J.A. Cabo, and T.B. Pedersen, "Inte- grating Data Warehouses with Web Data: A Survey," IEEE Trans. Knowledge and Data Eng., preprint, 21 Dec. 2007, doi:10.1109/TKDE.2007.190746.(PrePrint)
[15] R D Porsolt, Panconi, J Roux, M Altenbaumer, S. Hampe pharmacol, Biochem behavior 46 (1993)
[16] Shamim Akhter Design and Synthesis of some new derivatives of isonipecotamide as potential therapeutic agents In: PhD dissertation. (2003).
IJSER © 2015 http://www.ijser.org





