

International Journal of Scientific & Engineering Research, Volume 5, Issue 5, May-2014 1561
ISSN 2229-5518
Chemistry Dept. College of Science Thi-Qar University
The present study investigate the effect of polyphenolic extracts of Capparis Spinosa L. leaves on some biochemical parameters in female rats in vitro. The work involved the following studies: Analytical study: involved extraction, isolation and identification of polyphenolic content of Capparis Spinosa L. extract. The UV-Vis Spectra and high performance liquid chromatography of extracted polyphenols are proved the presence of (Gallic acid,Caffeic acid, Coumaric acid , Vanillic acid , Syringic acid, Ferulic acid, Chlorogenic acid, Rutin and Quercetin) in the extract. Biochemical parameters: all rats in bio-chemical parameters are divided into 4 groups, group (A) positive control (normal) that were treated with distill water (D.W), negative control group(B) that were treated with alloxan (125mg/kg) B.W. only diabetes rats that were treated with (15 mg/kg) B.W. of Capparis spinosa L. extract for 4 weeks, and group (D) alloxan-induced diabetes rats that were treated with (25 mg/kg) B.W. of Capparis spinosa L. extract for (4 weeks). The results indicated that group (B) showed a significant increase (P<0.01) in serum glucose compared with control group (A). The rats were fed with plant extract groups(C and D) show a significant decrease (P<0.01) in the level of serum glucose in comparison to diabetic animal group(B).
Diabetes mellitus is a disease commone in all partes of the word (Anonymous, 2007). It is defined as a group of disorders characterized by hyperglycemia, altered metabolism of lipids, carbohydrates and proteins (Patel et al., 2011; Warjeet, 2011). Chronic hyperglycemia is considered a major initiator of diabetic complications (Rama et al.,
2009) ,and causes damage to eyes, kidneys, nerves, heart and blood vessels (Mayfield,
1998). It is becoming the third “killer” of the health of mankind along with cancer cardiovascular and cerebrovascular diseases (Chauhan et al., 2010). Diabetes mellitus is caused by the abnormality of carbohydrate metabolism which is linked to low blood insulin level or insensitivity of target organs to insulin (Maiti et al., 2004). The increasing proportion of the aging population, consumption of calorie rich diet, obesity and sedentary lifestyle have led to a tremendous increase in the number of diabetics worldwide (Vats et al., 2004). The word “diabetes” is derived from the Greek word “Diab” (meaning to pass through, referring to the cycle of heavy thirst and frequent urination); “mellitus is the Latin word for “sweetened with honey” (refers to the presence of sugar in the urine) (Warjeet Singh, 2011). Diabetes is usually accompanied by increased production of free radicals or
impaired antioxidant defenses (Memisogullari and Bakan, 2004).
IJSER © 2014 http://www.ijser.org

International Journal of Scientific & Engineering Research, Volume 5, Issue 5, May-2014 1562
ISSN 2229-5518
The herbal drugs with antidiabetic activity are yet to be commercially formulated as modern medicines, even though they have been acclaimed for their therapeutic properties in the traditional systems of medicine (Wadkar et al., 2008). The World Health Organisation not only encourages the use of plant medicines, but also recommended scientific evaluation of the hypoglycaemic properties of plant extracts (Dirks, 2004).
Phenolic compounds are found in almost every plant-derived food, Fruits and vegetables contain several thousand structurally diverse phytochemicals, of which a large fraction are polyphenols (Scalbert and Williamson, 2000). The beneficial effects of polyphenols are mainly attributed to their antioxidant properties, many dietary polyphenols are known antioxidants (Surh, 2003; Pan and Ho, 2008; Miller and Ruiz- Larrea, 2002). Polyphenols exhibit multiple pharmacological properties such as anti- microbial, anti-allergenic, anti-ulcerogenic, anti-neo plastic, and anti- inflammatory activities (Formica and Regelson, 1995). The effects of polyphenols therapeutically relevant for the biological systems, are: they reduce the scavenger properties for oxygen free radicals (Burns, 2000), they reduce platelet aggregability (Jeong et a .,
1999), and they decrease arterial blood pressure (Hiroko et al., 2004). The bioavailability and biological properties of dietary polyphenols vary to a great extent and depend on their chemical structure (Manach et al., 2005; Loke et al., 2008 ).
Capparis Spinosa L. plant
Capparis spinosa L. is well known with its common name ‘Capers’ in different countries (Azaizeh et al., 2003). This plant also known as the caper bush, is a perennial winter deciduous species that bears rounded, fleshy leaves and large white to pinkish flowers (Ramezani et al., 2008). Extracts of different parts of C. spinosa have been shown to possess biological activity against a large number of pathogens (Chopra et al.,1996). Antifungal ,antibacterial, anti-amoebic, and anti-worm activities have been demonstrated (Asolkar et al.,1992; Guba Bakshi et al.,1999), antidiabetic
,antihyperlipidemic (Eddouks et al., 2005), anti hypertensive, poultice (Baytop et al.,1984) antileishmania, antihepatotoxic, , and antiallergic activities (Jacobson and Schlein, 1999; Gadgoli et al.,1999 ; Trombetta et al.,2005).
Capparis spinosa L. leaves were collected in May- 2013 from Nasiriyah city at Iraq, then it was authenticated and specimen of plant was classified in biological department- college of science at university of Thi qar in Iraq by Asst. prof. Hayder Radhi . The leaves were cleaned , washed by distilled water, dried at room temperature for two weeks, ground
as powder and kept in Dark glass containers for further use.
IJSER © 2014 http://www.ijser.org

International Journal of Scientific & Engineering Research, Volume 5, Issue 5, May-2014 1563
ISSN 2229-5518
Chloroform, ethanol, sodium hydroxide, hydrochloric acid, ferric chloride, acetic acid, lead acetate, - naphthol, sulphuric acid, Potassium citrate, mercuric chloride, Potassium hydroxide, n-hexane.
(500 g) of the powder dry leaves were defatted by washing several times with hexane(1L) at (60°C) , then it was macerated with (800mL) of acetic acid (2% v/v), the mixture were placed in sterile conical flask volume (2000mL) and put in water bath(60°C) for 8h, then the extraction process done by reflex condenser. The mixture was heated at50
C and then left to cool. The suspension was filtered by Buchner funnel by Whatman no.1 filter paper and use vacuum pump. The precipitate was canceled and the filtrate volume was measured then n-propanol was added into filtrate with the same volume of filtrate. Then (NaCl) added until to become solution super saturated. Then, it was evaporator by using rotary evaporator until drying (Gayon, 1972).
Isolated polyphenols were underwent a number of different tests such as:
Mole, 1994).
2001)
Tannins : was achieved by using (1%) lead acetate (Molan et al., 1997). Carbohydrates test: was done by using Molish΄s reagent (Harborne, 1984). Glycosides test: was carried out by using Benedict΄ s reagent (Harborne, 1984). Alkaloids test: was done by using Wagner΄s reagent (Harborne, 1984).
The absorption spectra of plant constituents was measured in very dilute solution against a blank solvent by using an automatic recording spectrophotometer. The solvent was used for UV spectroscopy is water : the method was performed by using polyphenolic. The sample solutions absorbance (A), was recorded by measuring the range scan from
190nm to800nm on a double beam UV-VIS spectrophotometer (Ikbal, 2004).
The extract were separated on FLC ( Fast Liquid Chromatography) column, C-18, 3 µm particle size (50×4.6 mm ID), mobile phase were 0.1% acetic acid in Dionized water: acetonitrile (20:80V/V) using linear gradients from 0-100%B in 10 minutes, detection UV set at 275 nm, flow rate 1.5 mL/min, the sequences of the eluted material of the standard were as follow, each standard was 25 µg/mL. 1.0g of the sample was weighted, then
IJSER © 2014 http://www.ijser.org

International Journal of Scientific & Engineering Research, Volume 5, Issue 5, May-2014 1564
ISSN 2229-5518
dissolved in 10 mL HPLC methanol, the sample shaking and agitated in ultrasonic bath for
10 minutes, then concentrated by evaporating the solvent with stream of liquid N2 until reach 0.2 µm (supelco company cat No16534K) then 20 µL were injected on HPLC column. The concentration for each compound were quantitatively determined by comparison the peak area of the standard with that of the samples. The concentration for each compound were quantitatively determined by comparison the peak area of the standard with that of the samples. The separation occurred on liquid chromatography Shimadzu 10 AV-LC equipped with binary delivery pump model LC-10A Shimadzu, the eluted peaks were monitored by UV-Vis 10 A-SPD spectrophotometer (Ikbal, 2004).
Twenty healthy adult female rats weighing (100-169 g) of 9-10 weeks old were used in the present study. Animals were housed in the animal house of biology Dept. College of Education, Thi-Qar University. Experiments were achieved between September-2013 & October-2013. Animals were housed in iron boxes bedded with wooden chips. During the experimental period six animals were kept in each box and they were housed under standard laboratory conditions (12h light: 12h dark photoperiod (LD) at 22 ± 2 Cº and relative humidity 45-55% (Coskun et al, 2004). Animals were fed on standard rat pellet and tap water ad libitum. The standard pellet contains wheat 66.6%, soya 25.6%, and sun flower oil 4.4%, lime stone 1.5%, salt 0.63%, methionine 0.158%, choline chloride 0.062% and trace elements 0.05% (Krinke, 2000).
Experimental animals were divided into four groups (5 rats in each group) upon the following designed:
- Group A: positive control (normal) that were treated with distill water (D.W).
- Group B: negative control that were treated with alloxan (125mg/kg) B.W. only to induction of diabetes.
- Group C: alloxan-induced diabetes rats that were treated with (15 mg/kg) B.W. of
Capparis spinosa L extract for (4 weeks).
- Group D: alloxan-induced diabetes rabbits that were treated with (25 mg/kg) B.W. of
Capparis spinosa L extract for (4weeks).
Diabetes was introduced experimentally in female laboratory rats by withholding food for (12 hours) approximately by a single subcutaneous injection of (125 mg / kg B.W) of alloxan monohydrate dissolved in distill water immediately before injection. The controlled animals received distill water only (Nimenibo-Vadia, 2003). Alloxan treated animals were allowed to drink of D-Glucose 5% overnight to prevent the potentially fatal
hypoglycemia occurring as a result of massive insulin release following alloxan injection.
IJSER © 2014 http://www.ijser.org

International Journal of Scientific & Engineering Research, Volume 5, Issue 5, May-2014 1565
ISSN 2229-5518
After seven days of injection the animals showing signs of extreme fatigue and frequent urination readied infected with diabetes (Alarcon-Aguilara et al., 2002).
5mL of blood were drawn from each animal of experimental groups, by heart puncture method after 12 hours fast. Using 60 gauge syringes, the sample was transferred into clean tube, left at room temperature for 15 minutes for clotting, centrifuged at 3000 rpm for 15 minutes, and then serum was separated and kept in a clean tube in the refrigerator at 2-8ºC until the time of assay.
Enzymatic Colorimetric (GOD - POD) used to estimate the level of glucose in the blood serum. Sera were then collected and stored in freezer used in the determination of the levels of glucose. It is notable that all measurements were duplicated for each sample.
Statistical analysis was done using the software SPSS version 15.0; the results were expressed as mean ± standard deviations (mean ± SD). One way ANOVA-test was used to compare parameters in different studied groups. Pvalues (P < 0.01) were considered statistically significant.
Capparis spinosa showed contain a number of antioxidant phytochemicals such as flavonoids and other polyphenols (Aslanturk and Tulay, 2009).
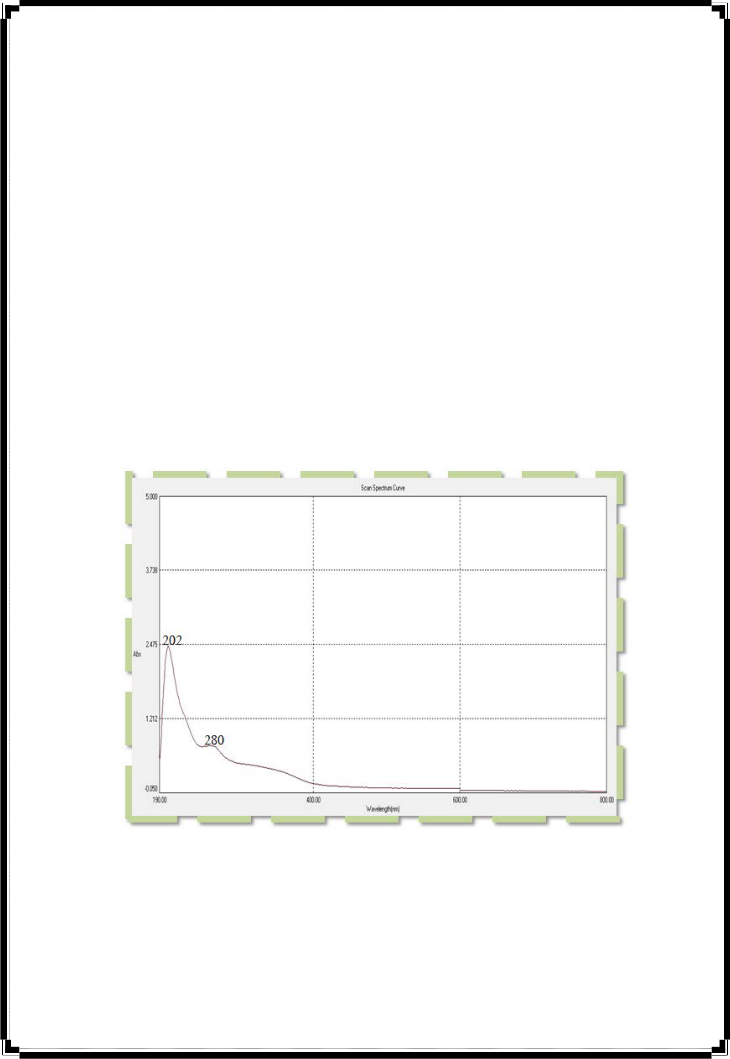
The chemical qualitative analysis tests results are shown in table (1) which indicate the presence of polyphenols, flavonoids and glycoside but the carbohydrate, tannins, saponins, alkaloids, terpinoids, terpenes and sterols gave a negative test, this ensure that polyphenolic compounds are pure. From table (1), it was found that glycosides because polyphenols is found normally connected with sacharids units . Figure (1) illustrates the UV-Vis spectra of the polyphenolic extract with max at (202nm) and another peak at
(280nm), these peak absorbed at (202) for π-π* electronic transitions due to the founding
of multi double bounds in aromatic rings for these compounds and another peak at (280) which is of low intensity appear in wave length longer than the first peak due to n- π* electronic transitions for non bonding electrons for oxygen atoms that are present in these
compounds.
IJSER © 2014 http://www.ijser.org
International Journal of Scientific & Engineering Research, Volume 5, Issue 5, May-2014 1566
ISSN 2229-5518
Table.1 Preliminary qualitative analysis tests of polypenols isolated from C. spinosa leaves
Reagent | Test result | Chemical Notes | Conclusions |
FeCl3(1%) | + | Formation of bluish green colour | Phenols are present |
Alchohol KOH (5N) | + | yellow precipitate | Flavonoids are absent |
Wagner | - | No reddish brown precipitate | Alkaloids are absent |
Pb(Ac)2 | - | No light brown precipitate | Tannins are absent |
Molish | - N | o Formation of violate ring | Carbohydrate are present |
Benedict | + | Formation of red precipitate | Glycosides are present |
HgCl2 | - | No white precipitate | Saponins are absent |
Conc. H2SO4 | - N | o purple red color | Terpinoids are absent |
Liebermann Burchard | - | No green colour | Terpenes and sterols are absent |
The plants extracts were analyzed to estimate their contents of polyphenol compounds. The identification of each compound was based on retention time in comparison with pure commercial standards (Shindalkar et al., 2005). The Figure (2) and fig (3) explained that the retention time of sample agrees with the retention time of the standard for most
contents in the extract.
IJSER © 2014 http://www.ijser.org

International Journal of Scientific & Engineering Research, Volume 5, Issue 5, May-2014 1567
ISSN 2229-5518
In this study and from HPLC results it was found that polyphenol extract contains some important compounds that include (gallic acid,caffeic acid, coumaric acid , Vanillic acid
(3) and the structures of these compounds were shown in table (2). The peaks of the mentioned chromatogram also pointed to presence of some unknown compounds that are thought represent derivatives of polyphenolic compounds.
IJSER © 2014 http://www.ijser.org

International Journal of Scientific & Engineering Research, Volume 5, Issue 5, May-2014 1568
ISSN 2229-5518
Figure.3 HPLC chromatogram of Capparis spinosa L phenolic extract.
seq | Polyphenolic contents in the standard | Retention time (min) | Area |
1 | Chlorogenic acid | 1.09 | 40854 |
2 | Caffeic acid | 1.77 | 55315 |
3 | Gallic acid | 2.49 | 44057 |
4 | Quercetin | 3.18 | 39424 |
5 | Syringic acid | 4.24 | 45691 |
6 | coumaric acid | 5.34 | 39763 |
7 | Ferulic acid | 6.09 | 37854 |
8 | Vanillic acid | 7.08 | 43141 |
9 | Rutin | 7.85 | 66993 |
IJSER © 2014 http://www.ijser.org

International Journal of Scientific & Engineering Research, Volume 5, Issue 5, May-2014 1569
ISSN 2229-5518
seq | Polyphenolic contents in the extract | Retention time (min) | Area | Conc. of polyphenolic compounds µg/mL |
1 | Chlorogenic acid | 1.08 | 22004 | 40.395 |
2 | Caffeic acid | 1.75 | 54240 | 73.542 |
3 | Gallic acid | 2.49 | 32976 | 56.136 |
4 | Quercetin | 3.16 | 76898 | 146.289 |
5 | Syringic acid | 4.22 | 121635 | 199.659 |
6 | P-coumaric acid | 5.33 | 45022 | 84.918 |
7 | Ferulic acid | 6.08 | 65018 | 128.82 |
8 | Vanillic acid | 7.09 | 34199 | 59.454 |
9 | Rutin | 7.83 | 29482 | 33.003 |
subject | Structure | Organizational name | ||
Chlorogenic acid | (1S,3R,4R,5R)-3-{[(2Z)-3-(3,4- dihydroxyphenyl)prop-2-enoyl]oxy}-1,4,5- trihydroxycyclohexanecarboxylic acid | |||
Caffeic acid | 3-(3,4-Dihydroxyphenyl)-2-propenoic acid | |||
Gallic acid | 3,4,5-trihydroxybenzoic acid | |||
Quercetin | 2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy- | |||
Quercetin | 4H-chromen-4-one | |||
Syringic acid | 4-hydroxy-3,5-dimethoxybenzoic acid | |||
P-Coumaric acid | 3-(4-hydroxyphenyl)-2-propenoic acid | |||
Ferulic acid | 3-(4-hydroxy-3-methoxy-phenyl)prop-2-enoic acid | |||
Vanillic acid | 4-Hydroxy-3-methoxybenzoic acid |
IJSER © 2014 http://www.ijser.org
International Journal of Scientific & Engineering Research, Volume 5, Issue 5, May-2014 1570
ISSN 2229-5518

Rutin
2-(3,4-dihydroxyphenyl)-5,7-dihydroxy-3-[α- L-rhamnopyranosyl-(1→6)-β-D- glucopyranosyloxy]-4H-chromen-4-one
Results of determination of serum glucose concentration in females rats groups (A , B
, C and D ) are given in table (5) and figure (4). A significant differences can be observed among (C and D) groups as compared with group (A) after treatment with (15 mg /kg B.W) and (25 mg /kg B.W) respectively of Capparis spinosa L extract for (4 weeks). Also there is a significant elevation that can be observed (P<0.01) in groups (B) as compared with group (A). The increase in blood glucose level is due to beta cell destruction by alloxan which lead to retardation of insulin production. Consequently, glucose can not enter into the cell leading to rise its level in the blood (Nelson and Cox, 2000 ; Decarvalho et al., 2003). The levels of serum glucose decreased significantly in (C and D) groups as compared to group (B) after treatment (4 weeks) days with (15 mg /kg B.W) and (25 mg /kg B.W) respectively. The antidiabetic activity of Capparis spinosa L. leaves may be due to the presence of polyphenoilc compounds that have hypoglycemic activity in the body (Lemhadri et al., 2007). Treatment with antioxidants may prevent or ameliorate abnormal function and biochemistry of nerve and protect nerves against to free radicals damage ( Karasu et al., 1995). Consequently it is postulated that these agents can prevent diabetic complications ( Montonen et al., 2004). Phenolic acids are well-known antioxidants, and also their antidiabetic activities have been reported in many studies( Mukherjee et al., 2006; Aslan et al., 2007). As that herbal extracts containing flavonoids was reported to demonstrate antidiabetic activity (Suba et al., 2004). On the basis of the above evidence, it is possible that the flavonoids present in this plant may be responsible for the observed antidiabetic activity.
Glucose conc mg/dl | ||
Group | No. | Mean±S.D |
A | 5 | 99.95± 3.34 ᵈ |
B | 5 | 203.37± 3.57 ᵃ |
C | 5 | 142.11± 2.58 ᵇ |
D | 5 | 128.24± 4.04 ᶜ |
L.S.D | 6.32 |
IJSER © 2014 http://www.ijser.org
International Journal of Scientific & Engineering Research, Volume 5, Issue 5, May-2014 1571
ISSN 2229-5518
250
200

150
100
50
0
A B C D
At the end of this thesis points below can be concluded:
- Polyphenolic extract isolated from Capparis spinosa L. leaves. in this study giving the immediately yields.
- Compounds that are exist in each extract identified by using UV-Visible spectrum, also appearance of several peaks indicating to the existing of phenolic acids.
- Compounds are determined through using HPLC technique, where in polyphenolic extract seven phenolic acids one flavonoid and one glycosides (Gallic acid,Caffeic acid, Coumaric acid , Vanillic acid , Syringic acid and Ferulic acid, Chlorogenic acid, Rutin and Quercetin) have been extracted from Capparis spinosa L. leaves.
- The extracted compounds were found to reduce serum glucose in alloxan induced diabetes mellituse females rats.
Alarcon-Aguilara, F. J. , Romas, R. , Perez-Gutierrez, S. , Aguilar-Contreras, A. , Contreras-Weber, C.C. and Flores-Saenz, J.L. (2002). Study of antihyperglycemic effect of plant used of antidiabetic. J. Ethnopharmacol. 61 (2) : 101 – 110.
Al-Assadi, I.( 2001). Study of hypoglycemia and antihyperglycemia action of Olea europae in animals and human, Ph.D. thesis, college of science, Basrah University, Iraq.
Anonymous.(2007).Diabetes mellitus statistics. American Heart Association .Feb, 18(1).
IJSER © 2014 http://www.ijser.org

International Journal of Scientific & Engineering Research, Volume 5, Issue 5, May-2014 1572
ISSN 2229-5518
Aslan, M. , Deliorman-Orhan, D. , Orhan, N. , Sezik, E. , Yesilada, E. (2007). In vivo antidiabetic and antioxidant potential of Helichrysumplicatum ssp. plicatumcapitulums in streptozotocin induced-diabetic rats. J Ethnopharmacol. 109:54–
9.
Aslanturk , O.S. and Tulay , A.c. , ( 2009 ). Genotoxic and Antimutagenic effect of
Capparis spinosa L. on the Allium cepa L. Root Tip Meristem Cells . Caryologia. 62(2)
: 114 – 123.
Asolkar, L.V. , Kakhar, K.K. , Chakre, O.J. (1992).Glossary of Indian medicinal plants with active principles. Part 1 Publications and information Directorate, New Delhi, India. pp. 166-7.
Azaizeh, H. , Fulder, S. , Khalil, K. , Said, O.(2003). Ethnomedicinal knowledge of local
Arab practitioners in the Middle East Region. Fitoterapia. 74: 98-108.
Baytop, P.(1984). Therapy with Medicinal Plants (Past and Present), Istanbul University
Publications, Istanbul.
Burns, J. (2000). Relationship among antioxidant activity, vasodilatation capacity, and phenolic content of red wine. J. Agric. Food Chem. 48:220-230.
Chauhan, A. , Sharma, P.K. , Srivastava, P. , Kumar, N. , Dudhe, R. (2010). Plants having potential anti-diabetic activity: A review. Der Pharmacia Lettre.2(3): 369-87.
Chopra, R.N. , Nayer, S.C. , Chopra, I,C, . (1996).Glossary of Indian medicinal plants.
National Institute of Science and Communication, New Delhi. 49-129.
Coskun, O. , Ocakci, A. , Bayraktaroglu, T. and Kanter, M. (2004). Exercise training prevents and protects streptozotocin-induced oxidative stress and beta-cell damage in rat pancreas. Tohoku J. Exp. Med. 203:145.
DeCarvalho, E.N. ; DeCarvalho, N.A.S. and Ferreiva, L.M. (2003). Experimental model of induction of diabetes mellitus in rats. Acta. Cir. Bras . 18.
Dirks, J.H.(2004). The drumbeat of renal failure: symbiosis of prevention and renal replacement therapy. Blood Purif . 22: 6–8.
Eddouks, M. , Lemhadri, A. , Michel, J.B. (2005). Hypolipidemic activity of aqueous extract of Capparis spinosa L. in normal and diabetic rats. Journal of Ethnopharmacology .98: 345–350
Formica, J.V., & Regelson, W. (1995). Review of the biology of quercetin and related biflavonoids. Food Chemistry Toxicology. 33: 1061–1080.
Gadgoli, C. , S.H. , Mishra. (1999). Antihepatotoxic activity of p-methoxy benzoic acid from Capparis spinosa. Journal of Ethnopharmacology. 66 :187-192.
Gayon, T.A. (1972). Plant phenolic. Oliver and Boyed, Edinboura . pp 254.
Guba Bakshi, D.N. , Sensarma, P. , Pal, D.C.(1999). A lexicon of medicinal plants of
India. Vol. 1, Naya Prakash, Calcutta, India. 360-5.
IJSER © 2014 http://www.ijser.org

International Journal of Scientific & Engineering Research, Volume 5, Issue 5, May-2014 1573
ISSN 2229-5518
Harbone, J.B. (1984). Phytochemical methods agude to moden techniques of plant analysis.2nd (edn) champman and hall, London, NewYork.
Hiroko, N. , Jin-Wen, X. , Katsumi, I. , Yasuo, N. ,Yukio, Y.(2004). Black and green tea polyphenols attenuate blood pressure increases in stroke – prone spontaneously hypertensive rats. J. Nutr. 134: 38 – 42.
Ikbal, F. A. (2004). Preparation of natural anticarcinogenic compounds and their effects on prostate tumor. Master, thesis. Collage engineering and science. Iraq. P, 51. in humans. Am. J. Clin. Nutr. 66: 1125.
Jacobson, R.L. ,Y. Schlein.(1999). Lectins and toxins in the plant diet of Phlebotomus papatasi (Diptera: Psychodidae) can kill Leishmania major promastigotes in the sandfly and in culture. Annals of Tropical Medicine and Parasitology .93 : 351-356.
Karasu, C. , Dewhurst, M. , Stevens, E.J. , Tomlinson, D.R. (1995). Effects of anti-oxidant treatment on sciatic nerve dysfunction in streptozotocin-diabetic rats: Comparison with essential fatty acids. Diabetologia. 38:129–34.
Krinke, G. J. (2000). The laboratory Rat. Academic press. A Harcourt Science and
Technology company Sand Diego.
Lemhadri, A. , Eddouks, M. , Sulpice, T. , and Burcelin R.(2007). Anti-hyperglycaemic and anti-obesity Effects of Capparisspinosa and Chamaemelumnobile aqueous extracts in HFD mice. Am J Pharmacol Toxicol. 2(3): 106-110.
Loke, W.M. , Proudfoot, J.M. , Stewart, S. (2008). Metabolic transformation has a profound effect on anti-inflammatory activity of flavonoids such as quercetin: lack of association between antioxidant and lipoxygenase inhibitory activity. Biochem Pharmacol. 75:1045–53.
Maiti R. , Jana, D. , Das, U.K. , Ghosh, D.(2004).Antidiabelic effect of aqueous extract of seed of tamarindus indicain streptozotocin induced diabetic rats. J Ethnopharmacol. 92:
85-91.
Manach, C. , Williamson, G. , Morand, C. , Scalbert, A. , Remesy, C.(2005).
Bioavailability and bioefficacy of polyphenols in humans. I. Review of 97 bioavailability studies. Am J Clin Nutr .81: 230S–42S.
Mayfield, J.(1998). Am. Fam. Physician .58: 1355.
Memisogullari, R. , and Bakan, E. (2004). Levels of ceruloplasmin, transferrin and lipid peroxidation in the serum of patients diabetes mellitus. J Diabetes Complications. 18:
193–7.
Miller, N.J. and Ruiz-Larrea, M.B.(2002). Flavonoids and other plant. J Nutr Environ Med
.12:39-51
Molan, A. , Mcnebb, W. , Attowd, G. , Min, B. , Peters, J. and Barry, T. (1997). The effect of condensed tannins from lotus species protein degradation and bacterial growth in the
rumen. J.Nat.Pro.Soc. 22:246.
IJSER © 2014 http://www.ijser.org

International Journal of Scientific & Engineering Research, Volume 5, Issue 5, May-2014 1574
ISSN 2229-5518
Montonen, J. , Knekt, P. , Järvinen, R. , Reunanen, A. (2004). Dietary antioxidant intake and risk of type 2 diabetes.Diabetes Care. 27:362–6.
Mukherjee, P.K. , Maiti, K. , Mukherjee, K. , Houghton, P.J. (2006). Leads from Indian medicinal plants with hypoglycemic potentials. J Ethnopharmacol. 106:1–28.
Nelson, D. L. and Cox, M.M. (2000). Lehninger Principles of Biochemistry. 3ed ed. Worth
Publishers. U.S.A. Pp : 790-885.
Nimenibo-Vadia, R. (2003). Control of hyperlipidemia hypercholesrolemia and hyperketonemia by aqueous extract of Dioscorea dumetorum tuber. Tropical journal of pharmaceutical Research, 2: 183-189.
Pan, M.H. ,Ho, C.T.(2008). Chemopreventive effects of naturaldietary compounds on cancer development. Chem Soc Rev 37:2558-74.
Patel, D.K. , Kumar, R. ,Prasad, S.K. ,Sairam, K. ,Hemalatha, S.(2011). Antidiabetic and in vitro antioxidant potential of Hybanthus enneaspermus (Linn) F. Muell in streptozotocin-induced diabetic rats. Asian Pac J Trop Biomed . 1(4): 316-22.
Rama, Srivatsan, MSc, et al.(2009). Antioxidants and Lipid Peroxidation with and without
Complications.12(12):121-127.
Ramezani, Z. , Aghel, V. ,Keyghobadi, H.(2008). Routine from different parts of Capparis spinosagrowing wild in Khuzastan and Iran. Pakistan J Biol Sci. 11(5): 768-72.
Scalbert, A. , Williamson, G.(2000). Dietary intake and bioavailability of polyphenols. J Nutr .130:2073S– 85S.
Shindalkar, S. , Madje, B. , Shingare, M.(2005). Liquid chromatographic retention and separation of phenols and related aromatic compounds on reversed phase columns. Chem. Soc. 49 (4) 377.
Suba, V. , Murugesan, T. , Bhaskara Rao, R. , Ghosh, L. , Pal, M. , Mandal, S.C. , et al.(2004). Antidiabetic potential ofBarleria lupulina extract in rats. Fitoterapia. 75:1–
4.
Surh YJ.(2003). Cancer chemoprevention with dietary phytochemicals. Nat Rev Cancer.
3:768-80.
Trombetta, D. , F. , Occhiuto, D. , Perri, C. , Puglia, N.A. , Santagati, A. , De Pasquale.(2005). Antiallergic and antihistaminic effect of two extracts of Capparis spinosa L. flowering buds. Phytotherapy Research. 19 (1) : 29-33.
Vats, V. , Yadav, S.P. , Grover, J.K.(2004). Ethanolic extract of Ocimum sanctum leaves partially attenuates streptozotocin-induced alterations in glycogen content and carbohydrate metabolism in rats. J Ethnopharmacol .90:155-160.
Wadkar, K.A. , Magdum, C.S. , Patil, S.S. , Naikwade, N.S.(2008). Antidiabetic potential and Indian medicinal plants. J Herbal Med and Toxicol .2:45-50.
IJSER © 2014 http://www.ijser.org

International Journal of Scientific & Engineering Research, Volume 5, Issue 5, May-2014 1575
ISSN 2229-5518
Warjeet Singh, L.(2011). Traditional medicinal plants of Manipur as antidiabetics. J Med
Plants Res. 5(5): 677-87.
Warjeet Singh, L.(2011). Traditional medicinal plants of Manipur as antidiabetics. J Med
Plants Res. 5(5): 677-87.
Waterman, P., and Mole, S.(1994). Analysis of phenolic plant metabolites. J. Plant
Medica. 55:1.
IJSER © 2014 http://www.ijser.org